Disclaimer: This content is for informational purposes only and should not replace professional medical advice.
The Mystery of Senile “Forgetfulness”
For generations, we have accepted experiences such as forgetting where we left our keys, struggling to recall an acquaintance’s name, or feeling that our reasoning lacks its former agility as inevitable. Science and society have conventionally viewed cognitive decline as a one-way street—a natural and irreversible wear and tear of the human “engine.” However, pioneering research from the University of California, San Francisco (UCSF), recently published in the prestigious journal Nature Aging, is turning this concept on its head. The data suggest that memory aging may not be a predetermined fate but a biological process that we can, in fact, rewind.
The Invisible “Villain”: The Discovery of the FTL1 Protein in the Aging Brain
In investigating what truly changes in the brain over the years, UCSFscientists conducted an exhaustive mapping of genes and p
roteins in the hippocampus—the brain’s “headquarters” for learning and memory. The result was surprising: among the thousands of molecules tracked, FTL1 (a protein associated with iron storage and metabolism) was the only one that stood out as consistently different between young and aged brains.
To understand its role, imagine the brain as a metropolis that relies on rigorous resource management. FTL1 is part of the ferritin complex, responsible for maintaining long-term iron balance. With aging, levels of this protein increase unchecked, acting as a chemical “limiter” that overloads cells and weakens neural connections in the aging brain.
The Science of Reversal: What the Studies Revealed
Definitive proof came from meticulous experiments with animal models. Researchers tested the influence of FTL1 on two distinct fronts:
Acceleration in Young Subjects: By artificially increasing FTL1 levels in young mice, scientists observed that their brains began to behave like those of older subjects, with a clear deterioration of memory.
Recovery in Elderly Subjects: The most impactful result occurred when FTL1 was reduced in old animals. They not only stopped losing memory but regained cognitive capacity, performing in maze and recognition tests comparably to young animals.
A crucial detail for scientific precision: researchers noted that while memory was restored, motor skills and anxiety levels remained unchanged. This proves that FTL1 is a surgical target, specifically affecting synaptic functions and memory, without causing a generalized and uncontrolled alteration in behavior. This is a key insight for understanding the aging brain.
“It’s truly a reversal of deficits,” states Dr. Saul Villeda, PhD and associate director of the UCSF Bakar Aging Research Institute. “We’re talking about something much more profound than just delaying or masking symptoms; it’s about restoring lost function.”
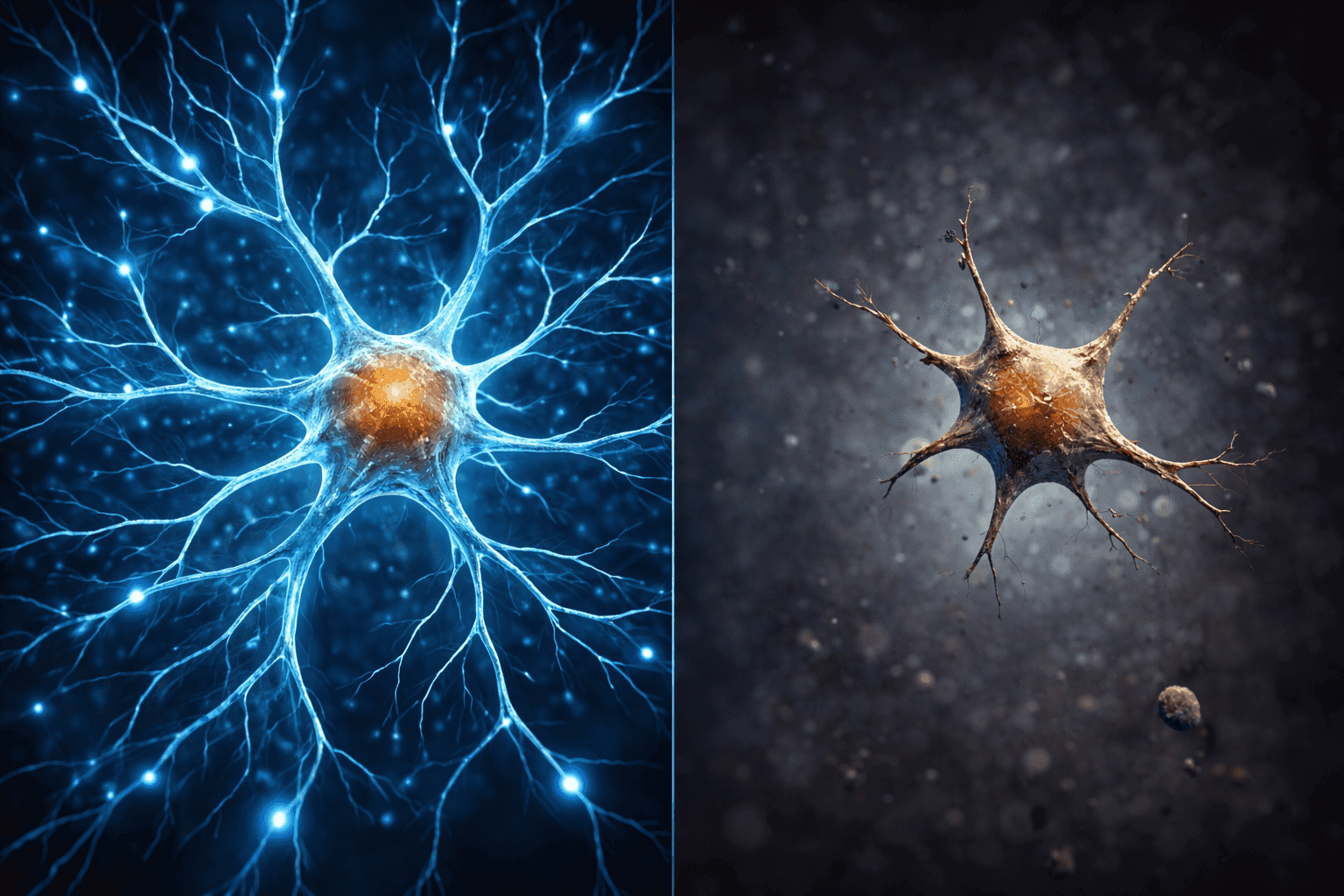
Why Connections Fail: The Visual Impact on Synapses in the Aging Brain
This discovery is not merely theoretical; it is visible under the microscope. In a young brain, neurons resemble a lush forest, with branched and complex networks that allow information to jump from one cell to another with speed.
Under excessive FTL1, this architecture collapses. Neurons lose their branches and become “single, simplified extensions.” The image is that of a vibrant forest transformed into a tangle of disconnected telephone wires and dry branches. This structural simplification is what makes it physically impossible for the aging brain to form new memories or access old ones efficiently. By reducing FTL1, the “forest” regrows, restoring the complexity of the branches.
The Metabolic Link: Energy for the Aging Brain
The research also revealed a deep connection between FTL1 and how the brain utilizes energy. High levels of the protein make cellular metabolism in the hippocampus slow and inefficient, like a battery that can no longer hold a charge.
Interestingly, scientists were able to prevent FTL1 damage using compounds that boost cellular metabolism. For us, this reinforces a vital lesson in preventive health: maintaining healthy metabolism through pillars such as balanced nutrition and physical exercise is not just an aesthetic or cardiovascular matter. It is a molecular strategy to protect neurons against energy failure caused by iron accumulation in the aging brain.
The Future of Cognitive Health: Real Perspectives for the Aging Brain
While we are facing a milestone that could revolutionize the treatment of diseases like Alzheimer’s and Parkinson’s, science proceeds with caution.
The Expert’s View
Dr. Andrew Steele, renowned anti-aging researcher, reflects on the next steps: “This is fascinating initial research, but there are steps to climb before we have drugs on the shelves. The work was done in animal models, and we need to validate whether the FTL1 protein plays exactly the same ‘aging driver’ role in humans.”
The next major challenge for regenerative medicine will be to develop therapies that can safely block FTL1 in humans, possibly through pharmacological or genetic interventions that mimic the results seen in the laboratory.
A New Perspective on Aging and the Brain
We are witnessing a paradigm shift. Brain aging is ceasing to be an inevitable fate to become a treatable and, perhaps, reversible condition. The discovery of FTL1 provides us with proof that the brain possesses extraordinary resilience, capable of rebuilding bridges we thought were destroyed by time.
For decades, brain aging was treated as inevitable. Findings like this suggest it may be, at least in part, a process we can influence.
This article is for informational purposes only and is not a substitute for professional medical advice, diagnosis, or treatment. Always consult a qualified healthcare provider before making any decisions about your health or starting a new regimen.
If this post saved you time, money, or prevented questionable life choices… you can tip below.